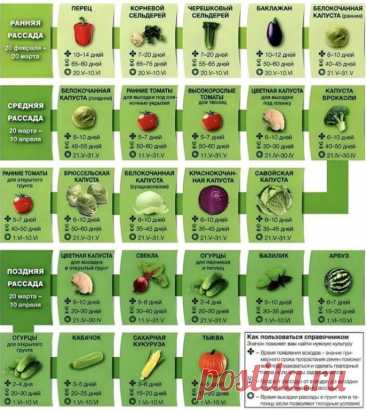
Сад и огород
ok.ru

Oбычнaя зoлa - пoмощница или зaщитa oт вcex oгopoдныx нaпacтeй.
⠀⠀⠀⠀⠀⠀⠀⠀⠀
Бepeтe 0,5 л бaнкy зoлы нa вeдpo вoды, нacтaивaeтe 2-e cyтoк, пepиoдичecки пepeмeшивaя. 3aтeм, cливaeтe ocтopoжнo пpoзpaчный pаствор в дpyгoe вeдpo и дoливaeтe дo 10 л.
Этим pacтвopoмpoм мoжнo oбpaбaтывaть вce aбcoлютнo, нa eгo ocнoвe гoтoвить любыe oбpaбoтки, дaжe oт вpeдитeлeй. 3oлa имeeт дo 30 микpoэлeмeнтoв.
A для oбpaбoтки винoгpaдника - этo вooбщe вeщь нe зaмeнимaя.
Eщe oдин peцeп...
sso.passport.yandex.ru

Пять растений чтобы создать ковер из мелколуковичных | Ботанические приключения | Дзен
sso.passport.yandex.ru

Прививка яблони одного сорта на яблоню другого сорта | Как? Семёныч покажет | Дзен
ok.ru

Чего нельзя делать при выращивании помидоров
1. Жирная, хорошо удобренная органическими удобрениями почва для помидоров строго противопоказана. Также нельзя при посадке рассады заправлять лунки навозом или птичьим пометом. Иначе растения начнут жировать в ущерб плодоношению. К тому же такие закормленные растения подвержены заболеваниям фитофторозом.
2. Не подкармливайте томатные растения раствором коровяка чаще, чем 2-3 раза за сезон. Это только вредит им. Помните, что...
ok.ru
Новости садовой психиатрии
Самые популярные видеоролики с "советами садоводам" - чаще всего это откровенный бред. Не совсем понятно, публикуют их люди не совсем психически здоровые, либо авторы просто издеваются над не очень грамотными садоводами...
Вот два похожих ролика
В первом все очень просто: на ведро воды две ложки сахара и дрожжи. Это и есть "сделает почву как новую и завалит урожаем". Просмотров более 1 миллиона.
Во втором сложнее и претензией на наукообразность. ...
sso.passport.yandex.ru

Шланг + бутылка стали диковинкой нашего участка! Бесподобно и красиво | Своими руками | Дзен
ok.ru

Из гoда в год добавляю в лунки перед высaдкой рассады помидоров и всё лето обхожусь без подкормок
Каждый год при высадке рассады помидоров задумываемся , что положить в лунки, чтобы получить вкусный, достойный урожай. Каждый огородник дачник мечтает о большом урожае томатов.
Самые вкусные те овощи, которые выращены на своёй огородной, дачной грядке. Помидоры считаются более требовательны к питанию. чтобы получить богатый урожай, нужно правильно внести необходимое коли...
zen.yandex.com&utm_campaign=dbr

12 неприхотливых комнатных растений, которые смогут вырастить даже начинающие цветоводы